Air pollution from a World War II wartime production plant
Air pollution is the introduction into the atmosphere of chemicals, particulates, or biological materials that cause discomfort, disease, or death to humans, damage other living organisms such as food crops, or damage the natural environment or built environment.
The atmosphere is a complex dynamic natural gaseous system that is essential to support life on planet Earth. Stratospheric ozone depletion due to air pollution has long been recognized as a threat to human health as well as to the Earth’s ecosystems.
Indoor air pollution (see Airlog) and urban air quality are listed as two of the World’s Worst Toxic Pollution Problems in the 2008 Blacksmith Institute World’s Worst Polluted Places report.
Pollutants
Main articles: Pollutant and Greenhouse gas
Before flue-gas desulfurization was installed, the emissions from this power plant in New Mexico contained excessive amounts of sulfur dioxide.
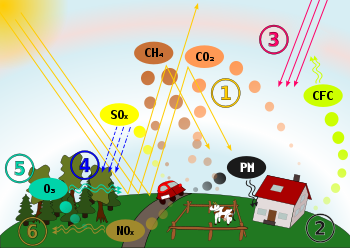
Schematic drawing, causes and effects of air pollution: (1) greenhouse effect, (2) particulate contamination, (3) increased UV radiation, (4) acid rain, (5) increased ground level ozone concentration, (6) increased levels of nitrogen oxides.
A substance in the air that can be adverse to humans and the environment is known as an air pollutant. Pollutants can be in the form of solid particles, liquid droplets, or gases. In addition, they may be natural or man-made. Pollutants can be classified as primary or secondary. Usually, primary pollutants are directly produced from a process, such as ash from a volcanic eruption, the carbon monoxide gas from a motor vehicle exhaust or sulphur dioxide released from factories. Secondary pollutants are not emitted directly. Rather, they form in the air when primary pollutants react or interact. An important example of a secondary pollutant isground level ozone — one of the many secondary pollutants that make up photochemical smog. Some pollutants may be both primary and secondary: that is, they are both emitted directly and formed from other primary pollutants.
Major primary pollutants produced by human activity include:
- Sulphur oxides (SOx) – especially sulphur dioxide, a chemical compound with the formula SO2. SO2 is produced by volcanoes and in various industrial processes. Since coal and petroleum often contain sulphur compounds, their combustion generates sulfur dioxide. Further oxidation of SO2, usually in the presence of a catalyst such as NO2, forms H2SO4, and thus acid rain.[2] This is one of the causes for concern over the environmental impact of the use of these fuels as power sources.
- Nitrogen oxides (NOx) – especially nitrogen dioxide are expelled from high temperature combustion, and are also produced naturally during thunderstorms byelectric discharge. Can be seen as the brown haze dome above or plume downwind of cities. Nitrogen dioxide is the chemical compound with the formula NO2. It is one of the several nitrogen oxides. This reddish-brown toxic gas has a characteristic sharp, biting odor. NO2 is one of the most prominent air pollutants.
- Carbon monoxide (CO)- is a colourless, odorless, non-irritating but very poisonous gas. It is a product by incomplete combustion of fuel such as natural gas, coal or wood. Vehicular exhaust is a major source of carbon monoxide.
- Volatile organic compounds – VOCs are an important outdoor air pollutant. In this field they are often divided into the separate categories of methane (CH4) and non-methane (NMVOCs). Methane is an extremely efficient greenhouse gas which contributes to enhanced global warming. Other hydrocarbon VOCs are also significant greenhouse gases via their role in creating ozone and in prolonging the life of methane in the atmosphere, although the effect varies depending on local air quality. Within the NMVOCs, the aromatic compounds benzene, toluene and xylene are suspected carcinogens and may lead to leukemia through prolonged exposure. 1,3-butadiene is another dangerous compound which is often associated with industrial uses.
- Particulates, alternatively referred to as particulate matter (PM), atmospheric particulate matter, or fine particles, are tiny particles of solid or liquid suspended in a gas. In contrast, aerosol refers to particles and the gas together. Sources of particulates can be man made or natural. Some particulates occur naturally, originating from volcanoes, dust storms, forest and grassland fires, living vegetation, and sea spray. Human activities, such as the burning of fossil fuels in vehicles, power plants and various industrial processes also generate significant amounts of aerosols. Averaged over the globe, anthropogenic aerosols—those made by human activities—currently account for about 10 percent of the total amount of aerosols in our atmosphere. Increased levels of fine particles in the air are linked to health hazards such as heart disease, altered lung function and lung cancer.
- Persistent free radicals connected to airborne fine particles could cause cardiopulmonary disease.
- Toxic metals, such as lead and mercury, especially their compounds.
- Chlorofluorocarbons (CFCs) – harmful to the ozone layer emitted from products currently banned from use.
- Ammonia (NH3) – emitted from agricultural processes. Ammonia is a compound with the formula NH3. It is normally encountered as a gas with a characteristic pungent odor. Ammonia contributes significantly to the nutritional needs of terrestrial organisms by serving as a precursor to foodstuffs and fertilizers. Ammonia, either directly or indirectly, is also a building block for the synthesis of many pharmaceuticals. Although in wide use, ammonia is both caustic and hazardous.
- Odors — such as from garbage, sewage, and industrial processes
- Radioactive pollutants – produced by nuclear explosions, nuclear events, war explosives, and natural processes such as the radioactive decay of radon.
Secondary pollutants include:
- Particulates created from gaseous primary pollutants and compounds in photochemical smog. Smog is a kind of air pollution; the word “smog” is a portmanteau of smoke and fog. Classic smog results from large amounts of coal burning in an area caused by a mixture of smoke and sulfur dioxide. Modern smog does not usually come from coal but from vehicular and industrial emissions that are acted on in the atmosphere by ultraviolet light from the sun to form secondary pollutants that also combine with the primary emissions to form photochemical smog.
- Ground level ozone (O3) formed from NOx and VOCs. Ozone (O3) is a key constituent of the troposphere. It is also an important constituent of certain regions of the stratosphere commonly known as the Ozone layer. Photochemical and chemical reactions involving it drive many of the chemical processes that occur in the atmosphere by day and by night. At abnormally high concentrations brought about by human activities (largely the combustion of fossil fuel), it is a pollutant, and a constituent of smog.
- Peroxyacetyl nitrate (PAN) – similarly formed from NOx and VOCs.
Minor air pollutants include:
- A large number of minor hazardous air pollutants. Some of these are regulated in USA under the Clean Air Act and in Europe under the Air Framework Directive.
- A variety of persistent organic pollutants, which can attach to particulates.
Persistent organic pollutants (POPs) are organic compounds that are resistant to environmental degradation through chemical, biological, and photolytic processes. Because of this, they have been observed to persist in the environment, to be capable of long-range transport, bioaccumulate in human and animal tissue, biomagnify in food chains, and to have potential significant impacts on human health and the environment.
Sources
Dust storm approaching Stratford, Texas
Controlled burning of a field outside ofStatesboro, Georgia in preparation for spring planting
Sources of air pollution refer to the various locations, activities or factors which are responsible for the releasing of pollutants into the atmosphere. These sources can be classified into two major categories which are:
Anthropogenic sources (man-made sources) mostly related to burning different kinds of fuel
- “Stationary Sources” include smoke stacks of power plants, manufacturing facilities (factories) and waste incinerators, as well as furnaces and other types of fuel-burning heating devices. In developing and poor countries, traditional biomass burning is the major source of air pollutants; traditional biomass includes wood, crop waste and dung.
- “Mobile Sources” include motor vehicles, marine vessels, aircraft and the effect of sound etc.
- Chemicals, dust and controlled burn practices in agriculture and forestry management. Controlled or prescribed burning is a technique sometimes used in forest management, farming, prairie restoration or greenhouse gas abatement. Fire is a natural part of both forest and grassland ecology and controlled fire can be a tool for foresters. Controlled burning stimulates the germination of some desirable forest trees, thus renewing the forest.
- Fumes from paint, hair spray, varnish, aerosol sprays and other solvents
- Waste deposition in landfills, which generate methane. Methane is highly flammable and may form explosive mixtures with air. Methane is also an asphyxiantand may displace oxygen in an enclosed space. Asphyxia or suffocation may result if the oxygen concentration is reduced to below 19.5% by displacement.
- Military, such as nuclear weapons, toxic gases, germ warfare and rocketry
Natural sources
- Dust from natural sources, usually large areas of land with little or no vegetation
- Methane, emitted by the digestion of food by animals, for example cattle
- Radon gas from radioactive decay within the Earth’s crust. Radon is a colorless, odorless, naturally occurring, radioactive noble gas that is formed from the decay of radium. It is considered to be a health hazard. Radon gas from natural sources can accumulate in buildings, especially in confined areas such as the basement and it is the second most frequent cause of lung cancer, after cigarette smoking.
- Smoke and carbon monoxide from wildfires
- Vegetation, in some regions, emits environmentally significant amounts of VOCs on warmer days. These VOCs react with primary anthropogenic pollutants—specifically, NOx, SO2, and anthropogenic organic carbon compounds—to produce a seasonal haze of secondary pollutants.
- Volcanic activity, which produce sulfur, chlorine, and ash particulates
Emission factors
Main article: AP 42 Compilation of Air Pollutant Emission Factors
Air pollutant emission factors are representative values that people attempt to relate the quantity of a pollutant released to the ambient air with an activity associated with the release of that pollutant. These factors are usually expressed as the weight of pollutant divided by a unit weight, volume, distance, or duration of the activity emitting the pollutant (e.g., kilograms of particulate emitted per tonne of coal burned). Such factors facilitate estimation of emissions from various sources of air pollution. In most cases, these factors are simply averages of all available data of acceptable quality, and are generally assumed to be representative of long-term averages.
There are 12 compounds in the list of POPs. Dioxins and furans are two of them and are intentionally created by combustion of organics, like open burning of plastics. The POPs are also endocrine disruptor and can mutate the human genes.
The United States Environmental Protection Agency has published a compilation of air pollutant emission factors for a multitude of industrial sources.[8] The United Kingdom, Australia, Canada and many other countries have published similar compilations, as well as the European Environment Agency.[10][11][12]
Indoor air quality (IAQ)
Main article: Indoor air quality
A lack of ventilation indoors concentrates air pollution where people often spend the majority of their time. Radon (Rn) gas, a carcinogen, is exuded from the Earth in certain locations and trapped inside houses. Building materials including carpeting and plywood emit formaldehyde (H2CO) gas. Paint and solvents give off volatile organic compounds (VOCs) as they dry. Lead paint can degenerate into dust and be inhaled. Intentional air pollution is introduced with the use of air fresheners,incense, and other scented items. Controlled wood fires in stoves and fireplaces can add significant amounts of smoke particulates into the air, inside and out.Indoor pollution fatalities may be caused by using pesticides and other chemical sprays indoors without proper ventilation.
Carbon monoxide (CO) poisoning and fatalities are often caused by faulty vents and chimneys, or by the burning of charcoal indoors. Chronic carbon monoxide poisoning can result even from poorly adjusted pilot lights. Traps are built into all domestic plumbing to keep sewer gas and hydrogen sulfide, out of interiors. Clothing emits tetrachloroethylene, or other dry cleaning fluids, for days after dry cleaning.
Though its use has now been banned in many countries, the extensive use of asbestos in industrial and domestic environments in the past has left a potentially very dangerous material in many localities. Asbestosis is a chronic inflammatory medical condition affecting the tissue of the lungs. It occurs after long-term, heavy exposure to asbestos from asbestos-containing materials in structures. Sufferers have severe dyspnea (shortness of breath) and are at an increased risk regarding several different types of lung cancer. As clear explanations are not always stressed in non-technical literature, care should be taken to distinguish between several forms of relevant diseases. According to theWorld Health Organisation (WHO), these may defined as; asbestosis, lung cancer, and Peritoneal Mesothelioma (generally a very rare form of cancer, when more widespread it is almost always associated with prolonged exposure to asbestos).
Biological sources of air pollution are also found indoors, as gases and airborne particulates. Pets produce dander, people produce dust from minute skin flakes and decomposed hair, dust mites in bedding, carpeting and furniture produce enzymes and micrometre-sized fecal droppings, inhabitants emit methane, mold forms in walls and generates mycotoxins and spores, air conditioning systems can incubateLegionnaires’ disease and mold, and houseplants, soil and surrounding gardens can produce pollen, dust, and mold. Indoors, the lack of air circulation allows these airborne pollutants to accumulate more than they would otherwise occur in nature.
Health effects
See also: Neuroplastic effects of pollution
Air pollution is a significant risk factor for multiple health conditions including respiratory infections, heart disease, and lung cancer, according to the WHO. The health effects caused by air pollution may include difficulty in breathing, wheezing, coughing, asthma and aggravation of existing respiratory and cardiac conditions. These effects can result in increased medication use, increased doctor or emergency room visits, more hospital admissions and premature death. The human health effects of poor air quality are far reaching, but principally affect the body’s respiratory system and the cardiovascular system. Individual reactions to air pollutants depend on the type of pollutant a person is exposed to, the degree of exposure, the individual’s health status and genetics.
The most common sources of air pollution include particulates, ozone, nitrogen dioxide, and sulfur dioxide. Both indoor and outdoor air pollution have caused approximately 3.3 million deaths worldwide. Children aged less than five years that live in developing countries are the most vulnerable population in terms of total deaths attributable to indoor and outdoor air pollution.
The World Health Organization states that 2.4 million people die each year from causes directly attributable to air pollution, with 1.5 million of these deaths attributable to indoor air pollution.”Epidemiological studies suggest that more than 500,000 Americans die each year from cardiopulmonary disease linked to breathing fine particle air pollution. . .” A study by the University of Birminghamhas shown a strong correlation between pneumonia related deaths and air pollution from motor vehicles. Worldwide more deaths per year are linked to air pollution than to automobile accidents. A 2005 study by the European Commission calculated that air pollution reduces life expectancy by an average of almost nine months across the European Union. Causes of deaths include aggravated asthma, emphysema, lung and heart diseases, and respiratory allergies. The US EPA estimates that a proposed set of changes in diesel engine technology (Tier 2) could result in 12,000 fewer premature mortalities, 15,000 fewer heart attacks, 6,000 fewer emergency room visits by children with asthma, and 8,900 fewer respiratory-related hospital admissions each year in the United States.[citation needed]
The US EPA estimates allowing a ground-level ozone concentration of 65 parts per billion, would avert 1,700 to 5,100 premature deaths nationwide in 2020 compared with the current 75-ppb standard. The agency projects the stricter standard would also prevent an additional 26,000 cases of aggravated asthma, and more than a million cases of missed work or school.
The worst short term civilian pollution crisis in India was the 1984 Bhopal Disaster. Leaked industrial vapours from the Union Carbide factory, belonging to Union Carbide, Inc., U.S.A., killed more than 25,000 people outright and injured anywhere from 150,000 to 600,000. The United Kingdom suffered its worst air pollution event when the December 4 Great Smog of 1952 formed over London. In six days more than 4,000 died, and 8,000 more died within the following months.[citation needed] An accidental leak of anthrax spores from a biological warfare laboratory in the former USSR in 1979 near Sverdlovsk is believed to have been the cause of hundreds of civilian deaths.[citation needed] The worst single incident of air pollution to occur in the US occurred in Donora, Pennsylvania in late October, 1948, when 20 people died and over 7,000 were injured.
A new economic study of the health impacts and associated costs of air pollution in the Los Angeles Basin and San Joaquin Valley of Southern California shows that more than 3800 people die prematurely (approximately 14 years earlier than normal) each year because air pollution levels violate federal standards. The number of annual premature deaths is considerably higher than the fatalities related to auto collisions in the same area, which average fewer than 2,000 per year.
Diesel exhaust (DE) is a major contributor to combustion derived particulate matter air pollution. In several human experimental studies, using a well validated exposure chamber setup, DE has been linked to acute vascular dysfunction and increased thrombus formation. This serves as a plausible mechanistic link between the previously described association between particulates air pollution and increased cardiovascular morbidity and mortality.
Effects on cardiovascular health
A 2007 review of evidence found ambient air pollution exposure is a risk factor correlating with increased total mortality from cardiovascular events (range: 12% to 14% per a 10 microg/m3 increase). PMID 19235364.
Air pollution is also emerging as a risk factor for stroke, particularly in developing countries where pollutant levels are highest. A 2007 study found that in women air pollution is associated not with hemorrhagic but with ischemic stroke. Air pollution was also found to be associated with increased incidence and mortality from coronary stroke in a cohort study in 2011. Associations are believed to be causal and effects may be mediated by vasoconstriction, low-grade inflammation or autonomic nervous system imbalance or other mechanisms.
Effects on cystic fibrosis
Main article: Cystic fibrosis
A study from around the years of 1999 to 2000, by the University of Washington, showed that patients near and around particulates air pollution had an increased risk of pulmonary exacerbations and decrease in lung function. Patients were examined before the study for amounts of specific pollutants like Pseudomonas aeruginosa or Burkholderia cenocepacia as well as their socioeconomic standing. Participants involved in the study were located in the United States in close proximity to an Environmental Protection Agency.[clarification needed] During the time of the study 117 deaths were associated with air pollution. Many patients in the study lived in or near large metropolitan areas in order to be close to medical help. These same patients had higher level of pollutants found in their system because of more emissions in larger cities. As cystic fibrosis patients already suffer from decreased lung function, everyday pollutants such as smoke, emissions from automobiles, tobacco smoke and improper use of indoor heating devices could further compromise lung function.
Effects on COPD and asthma
Main article: Chronic obstructive pulmonary disease
Chronic obstructive pulmonary disease (COPD) includes diseases such as chronic bronchitis and emphysema.
Researches have demonstrated increased risk of developing asthma and COPD from increased exposure to traffic-related air pollution. Additionally, air pollution has been associated with increased hosptializations and mortality from asthma and COPD.
A study conducted in 1960-1961 in the wake of the Great Smog of 1952 compared 293 London residents with 477 residents of Gloucester, Peterborough, and Norwich, three towns with low reported death rates from chronic bronchitis. All subjects were male postal truck drivers aged 40 to 59. Compared to the subjects from the outlying towns, the London subjects exhibited more severe respiratory symptoms (including cough, phlegm, and dyspnea), reduced lung function (FEV1 and peak flow rate), and increased sputum production and purulence. The differences were more pronounced for subjects aged 50 to 59. The study controlled for age and smoking habits, so concluded that air pollution was the most likely cause of the observed differences.
It is believed that much like cystic fibrosis, by living in a more urban environment serious health hazards become more apparent. Studies have shown that in urban areas patients suffer mucus hypersecretion, lower levels of lung function, and more self diagnosis of chronic bronchitis and emphysema.
Links to cancer
A review of evidence regarding whether ambient air pollution exposure is a risk factor for cancer in 2007 found solid data to conclude that long-term exposure to PM2.5 (fine particulates) increases the overall risk of nonaccidental mortality by 6% per a 10 microg/m3 increase.PMID 19235364
Exposure to PM2.5 was also associated with an increased risk of mortality from lung cancer (range: 15% to 21% per a 10 microg/m3 increase) and total cardiovascular mortality (range: 12% to 14% per a 10 microg/m3 increase). PMID 19235364
The review further noted that living close to busy traffic appears to be associated with elevated risks of these three outcomes (increase in lung cancer deaths, cardiovascular deaths, and overall nonaccidental deaths. PMID 19235364
The reviewers also found suggestive evidence that exposure to PM2.5 is positively associated with mortality from coronary heart diseases and exposure to SO2 increases mortality from lung cancer, but the data was insufficient to provide solid conclusions.
In 2011, a large Danish epidemiological study found an increased risk of lung cancer for patients who lived in areas with high nitrogen oxide concentrations. In this study, the association was higher for non-smokers than smokers. An additional Danish study, also in 2011, likewise noted evidence of possible associations between air pollution and other forms of cancer, including cervical cancer and brain cancer.
Effects on children
Around the world, children living in cities with high exposure to air pollutants are at increased risk of developing asthma, pneumonia and other lower respiratory infections. Because children are outdoors more and have higher minute ventilation they are more susceptible to the dangers of air pollution. Risks of low initial birth weight are also heightened in such cities.
The World Health Organization reports that the greatest concentrations of particulates are found in countries with low economic world power and high poverty and population growth rates. Examples of these countries include Egypt, Sudan, Mongolia, and Indonesia. However even in the United States, despite the passage of the Clean Air Act in 1970, in 2002 at least 146 million Americans were living in non-attainment areas—regions in which the concentration of certain air pollutants exceeded federal standards. These dangerous pollutants are known as the criteria pollutants, and include ozone, particulates, sulfur dioxide, nitrogen dioxide, carbon monoxide, and lead. Protective measures to ensure children’s health are being taken in cities such as New Delhi, India where buses now use compressed natural gas to help eliminate the “pea-soup” smog.
Health effects in relatively “clean” areas
Even in the areas with relatively low levels of air pollution, public health effects can be significant and costly, since a large number of people breathe in such pollutants. A 2005 scientific study for the British Columbia Lung Association showed that a small improvement in air quality (1% reduction of ambient PM2.5 and ozone concentrations) would produce a $29 million in annual savings in the Metro Vancouverregion in 2010. This finding is based on health valuation of lethal (death) and sub-lethal (illness) effects.
Reduction efforts
There are various air pollution control technologies and land use planning strategies available to reduce air pollution. At its most basic level land use planning is likely to involve zoning and transport infrastructure planning. In most developed countries, land use planning is an important part of social policy, ensuring that land is used efficiently for the benefit of the wider economy and population as well as to protect the environment.
Efforts to reduce pollution from mobile sources includes primary regulation (many developing countries have permissive regulations),[citation needed] expanding regulation to new sources (such as cruise and transport ships, farm equipment, and small gas-powered equipment such as lawn trimmers, chainsaws, and snowmobiles), increased fuel efficiency (such as through the use of hybrid vehicles), conversion to cleaner fuels (such as bioethanol, biodiesel, or conversion to electric vehicles).
Control devices
The following items are commonly used as pollution control devices by industry or transportation devices. They can either destroy contaminants or remove them from an exhaust stream before it is emitted into the atmosphere.
- Particulate control
- Mechanical collectors (dust cyclones, multicyclones)
- Electrostatic precipitators An electrostatic precipitator (ESP), or electrostatic air cleaner is a particulate collection device that removes particles from a flowing gas (such as air) using the force of an induced electrostatic charge. Electrostatic precipitators are highly efficient filtration devices that minimally impede the flow of gases through the device, and can easily remove fine particulates such as dust and smoke from the air stream.
- Baghouses Designed to handle heavy dust loads, a dust collector consists of a blower, dust filter, a filter-cleaning system, and a dust receptacle or dust removal system (distinguished from air cleaners which utilize disposable filters to remove the dust).
-
- Particulate scrubbersWet scrubber is a form of pollution control technology. The term describes a variety of devices that use pollutants from a furnace flue gas or from other gas streams. In a wet scrubber, the polluted gas stream is brought into contact with the scrubbing liquid, by spraying it with the liquid, by forcing it through a pool of liquid, or by some other contact method, so as to remove the pollutants.
Air pollutant concentrations
Air pollutant concentrations, as measured or as calculated by air pollution dispersion modeling, must often be converted or corrected to be expressed as required by the regulations issued by various governmental agencies. Regulations that define and limit the concentration of pollutants in the ambient air or in gaseous emissions to the ambient air are issued by various national and state (or provincial)environmental protection and occupational health and safety agencies.
Such regulations involve a number of different expressions of concentration. Some express the concentrations as ppmv (parts per million by volume) and some express the concentrations as mg/m3(milligrams per cubic meter), while others require adjusting or correcting the concentrations to reference conditions of moisture content, oxygen content or carbon dioxide content. This article presents methods for converting concentrations from ppmv to mg/m3 (and vice versa) and for correcting the concentrations to the required reference conditions.
All of the concentrations and concentration corrections in this article apply only to air and other gases. They are not applicable for liquids.
Converting air pollutant concentrations
The conversion equations depend on the temperature at which the conversion is wanted (usually about 20 to 25 °C). At an ambient sea level atmospheric pressure of 1 atm (101.325 kPa or 1.01325 bar), the general equation is:
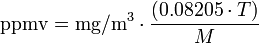
and for the reverse conversion:

| where: | |
| mg/m3 | = milligrams of pollutant per cubic meter of air at sea level atmospheric pressure and T |
| ppmv | = air pollutant concentration, in parts per million by volume |
| T | = ambient temperature in K = 273.15 + °C |
| 0.08205 | = Universal gas constant in atm·m3/(kmol·K) |
| M | = molecular mass (or molecular weight) of the air pollutant |
Notes:
- 1 atm = absolute pressure of 101.325 kPa or 1.01325 bar
- mol = gram mole and kmol = 1000 gram moles
- Pollution regulations in the United States typically reference their pollutant limits to an ambient temperature of 20 to 25 °C as noted above. In most other nations, the reference ambient temperature for pollutant limits may be 0 °C or other values.
- Although ppmv and mg/m3 have been used for the examples in all of the following sections, concentrations such as ppbv (i.e., parts per billion by volume), volume percent, mole percent and many others may also be used for gaseous pollutants.
- Particulate matter (PM) in the atmospheric air or in any other gas cannot be expressed in terms of ppmv, ppbv, volume percent or mole percent. PM is most usually (but not always) expressed as mg/m3of air or other gas at a specified temperature and pressure.
- For gases, volume percent = mole percent
- 1 volume percent = 10,000 ppmv (i.e., parts per million by volume) with a million being defined as 106.
- Care must be taken with the concentrations expressed as ppbv to differentiate between the British billion which is 1012 and the USA billion which is 109 (also referred to as the long scale and short scale billion, respectively).
Correcting concentrations for altitude
Air pollutant concentrations expressed as mass per unit volume of atmospheric air (e.g., mg/m3, µg/m3, etc.) at sea level will decrease with increasing altitude. The concentration decrease is directly proportional to the pressure decrease with increasing altitude. Some governmental regulatory jurisdictions require industrial sources of air pollution to comply with sea level standards corrected for altitude. In other words, industrial air pollution sources located at altitudes well above sea level must comply with significantly more stringent air quality standards than sources located at sea level (since it is more difficult to comply with lower standards). For example, New Mexico‘s Department of the Environment has a regulation with such a requirement.
The change of atmospheric pressure with altitude can be obtained from this equation:

Given an air pollutant concentration at sea-level atmospheric pressure, the concentration at higher altitudes can be obtained from this equation:

| where: | |
| h | = altitude, in km |
|---|---|
| P | = atmospheric pressure at sea level |
| Ph | = atmospheric pressure at altitude h |
| C |
= Air pollutant concentration, in mass per unit volume at sea level atmospheric pressure and specified temperature TCh= Concentration, in mass per unit volume at altitude h and specified temperature T
As an example, given an air pollutant concentration of 260 mg/m3 at sea level, calculate the equivalent pollutant concentration at an altitude of 2800 meters:
Ch = 260 × [ { 288 – (6.5)(2.8) } / 288] 5.2558 = 260 × 0.71 = 185 mg/m3
Note:
- The above equation for the decrease of air pollution concentrations with increasing altitude is applicable only for about the first 10 km of altitude in the troposphere (the lowest atmospheric layer) and is estimated to have a maximum error of about 3 percent. However, 10 km of altitude is sufficient for most purposes involving air pollutant concentrations.
Correcting concentrations for reference conditions
Many environmental protection agencies have issued regulations that limit the concentration of pollutants in gaseous emissions and define the reference conditions applicable to those concentration limits. For example, such a regulation might limit the concentration of NOx to 55 ppmv in a dry combustion exhaust gas (at a specified reference temperature and pressure) corrected to 3 volume percent O2 in the dry gas. As another example, a regulation might limit the concentration of total particulate matter to 200 mg/m3 of an emitted gas (at a specified reference temperature and pressure) corrected to a dry basis and further corrected to 12 volume percent CO2 in the dry gas.
Environmental agencies in the USA often use the terms “dscf” or “scfd” to denote a “standard” cubic foot of dry gas. Likewise, they often use the terms “dscm” or “scmd” to denote a “standard” cubic meter of gas. Since there is no universally accepted set of “standard” temperature and pressure, such usage can be and is very confusing. It is strongly recommended that the reference temperature and pressure always be clearly specified when stating gas volumes or gas flow rates.
Correcting to a dry basis
If a gaseous emission sample is analyzed and found to contain water vapor and a pollutant concentration of say 40 ppmv, then 40 ppmv should be designated as the “wet basis” pollutant concentration. The following equation can be used to correct the measured “wet basis” concentration to a “dry basis” concentration:
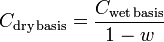
| where: | |
| C | = Concentration of the air pollutant in the emitted gas |
|---|---|
| w | = fraction, by volume, of the emitted gas |
As an example, a wet basis concentration of 40 ppmv in a gas having 10 volume percent water vapor would have a:
Cdry basis = 40 ÷ ( 1 – 0.10 ) = 44.4 ppmv.
Correcting to a reference oxygen content
The following equation can be used to correct a measured pollutant concentration in a dry emitted gas with a measured O2 content to an equivalent pollutant concentration in a dry emitted gas with a specified reference amount of O2:

| where: | |
| Cr | = corrected concentration of a dry gas with a specified reference volume % O2 |
|---|---|
| Cm | = measured concentration in a dry gas having a measured volume % O2 |
As an example, a measured NOx concentration of 45 ppmv in a dry gas having 5 volume % O2 is:
45 × ( 20.9 – 3 ) ÷ ( 20.9 – 5 ) = 50.7 ppmv of NOx
when corrected to a dry gas having a specified reference O2 content of 3 volume %.
Note:
- The measured gas concentration Cm must first be corrected to a dry basis before using the above equation.
Correcting to a reference carbon dioxide content
The following equation can be used to correct a measured pollutant concentration in an emitted gas (containing a measured CO2 content) to an equivalent pollutant concentration in an emitted gas containing a specified reference amount of CO2:

| where: | |
| Cr | = corrected concentration of a dry gas having a specified reference volume % CO2 |
|---|---|
| Cm | = measured concentration of a dry gas having a measured volume % CO2 |
As an example, a measured particulates concentration of 200 mg/m3 in a dry gas that has a measured 8 volume % CO2 is:
200 × ( 12 ÷ 8 ) = 300 mg/m3
when corrected to a dry gas having a specified reference CO2 content of 12 volume %.
[button size=”small” type=”warning” value=”Determination of Air Pollutants sox and nox” href=”#”]
Air Pollution Sampling and Analysis – Indian Institute of Technology
DETERMINING FACILITY EMISSIONS FOR TITLE V APPLICABILITY
Kansas Air Regulations Five_Steps_to_Determine_Whether_Your_Facility_Needs_an_Air_Permit
منبع : en.wikipedia.org
بازنشر » berroz.com